April 13, 2023
Sustainable Batteries for the Future
Research groups at ISTA strive to make energy-saving greener
Batteries are indispensable for global efforts to reduce fossil fuel use. However, challenges remain: their production requires a lot of energy, the materials used are rare, and batteries are difficult to recycle. Several research groups at the Institute of Science and Technology Austria (ISTA) are therefore working on new, more environmentally friendly, and more efficient batteries.

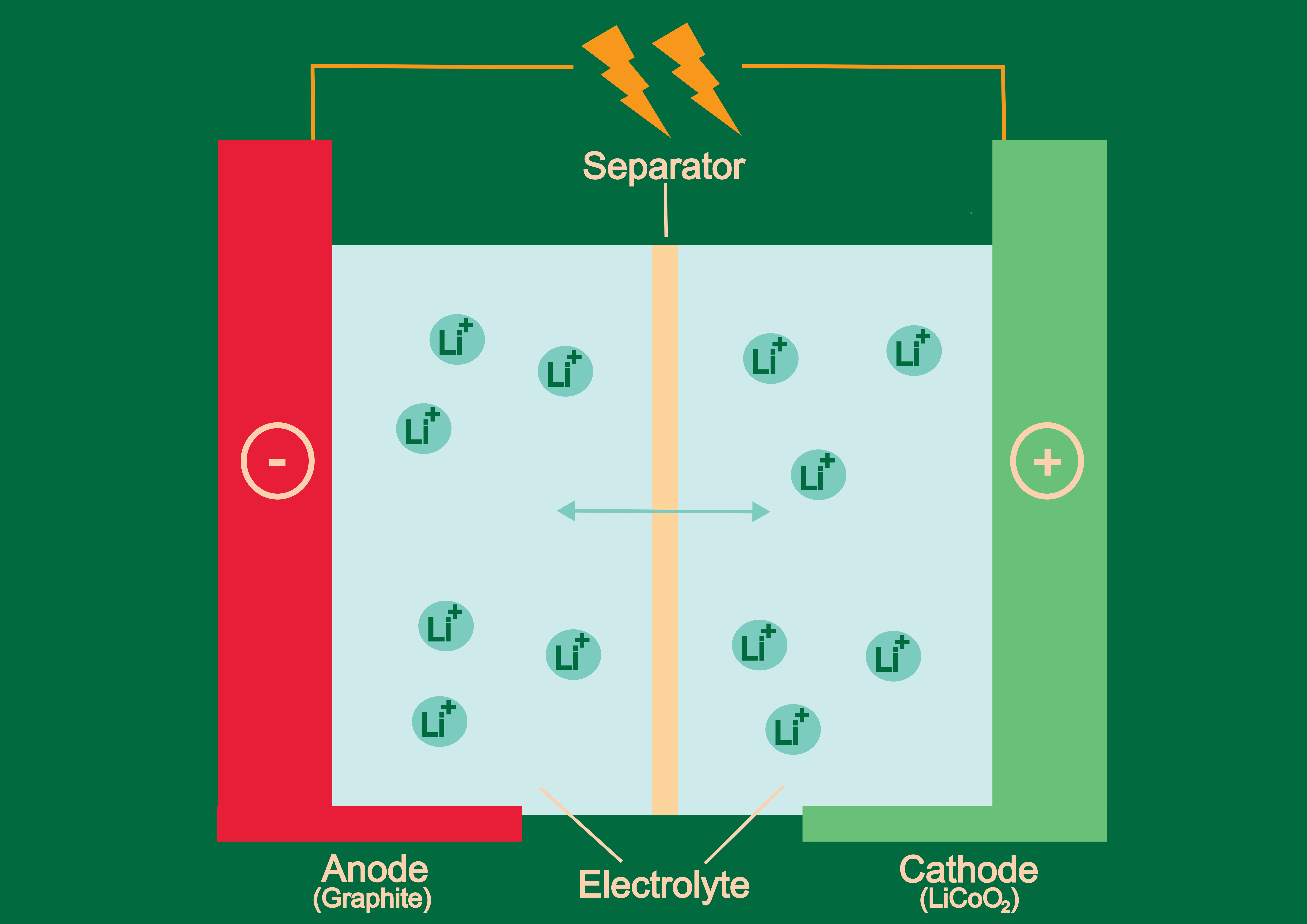
From cell phones to laptops to electric vehicles and beyond, lithium-ion (Li-ion) batteries are essential in the daily lives of many. Still, what seems like a boon, causes numerous problems: The metals used in the battery cells—mainly cobalt and lithium—can only be sourced in a few places. As a consequence, ecosystems are being destroyed, people toil under catastrophic working conditions, and the industry is dependent on a fragile supply chain, where a sudden cut-off could have drastic consequences.
Research groups in the fields of chemistry and physics at the Institute of Science and Technology Austria (ISTA) are trying to tackle these problems. Their projects all have the same goal: Finding novel materials to make batteries more sustainable.
Recycling through living nature

2021 marked the first year in which the cost of Li-ion batteries went up. “The reason is the scarcity of cobalt and lithium in combination with sky-rocketing demand, which are essential to Li-ion batteries in their current form,” says Stefan Freunberger, Assistant Professor at ISTA. The good news: You can replace the cobalt—exactly what Freunberger intends to do. His research group is focusing on making batteries as performant as Li-ion batteries, but only from very abundant elements. In particular, they use organic materials consisting only of elements like carbon, hydrogen, and oxygen, which derive—as the name suggests—from organic sources. The research group is also investigating the direct use of inorganic elements like sulfur.
Moreover, unlike cobalt-based ones, these materials have the potential to be recycled fully by living nature and the overall energy for battery production can be reduced. “Normally, the fabrication of Li-ion batteries consumes lots of energy and has a large CO2 footprint. By using the right materials, you can decrease these values by an order of magnitude,” says Freunberger. “If all goes well, we’ll have functional batteries with organic elements within a very few years. We are not yet ready for commercialization, but our results are promising,” Freunberger concludes. The researchers are supported in their ambitious project by an ERC Proof of Concept Grant, which helps to evaluate the commercial potential of research results.
Magnesium: A beacon of hope
One material that could be mined in an environmentally friendly way is magnesium. Additionally, it is one of the ten most common elements in the Earth’s crust. This shiny silver light metal is the focus of “MAGNIFICO”. Researchers from the Austrian Institute of Technology (AIT) and ISTA are joining forces in this Austrian Research Promotion Agency (FFG)-funded project. Together, they seek to use magnesium for the battery anode instead of lithium. However, though magnesium generally has the correct electrochemical properties, magnesium anodes are incompatible with most electrolytes—the medium that allows current to flow within the battery. In particular, the electrolyte decomposes in the presence of magnesium, thus restricting the electrical conductivity. To overcome this critical issue, the project team is working to establish a shield for the anode that prevents electrolyte decomposition. For her part in this ambitious project, ISTA Assistant Professor Bingqing Cheng and her research group provide computational calculations and machine learning to better understand and predict material properties.

Industrial batteries reimagined
While Li-ion batteries are mainly installed in the countless electrical appliances of our everyday life, the research group of VERBUND Professor of Energy Science Maria Ibáñez at ISTA is thinking bigger. “Industrial companies have the highest energy demand”, explains Mario Palacios Corella, postdoc in the Ibáñez group. “Our goal is to power them with a new, more sustainable, redox flow battery technology.”
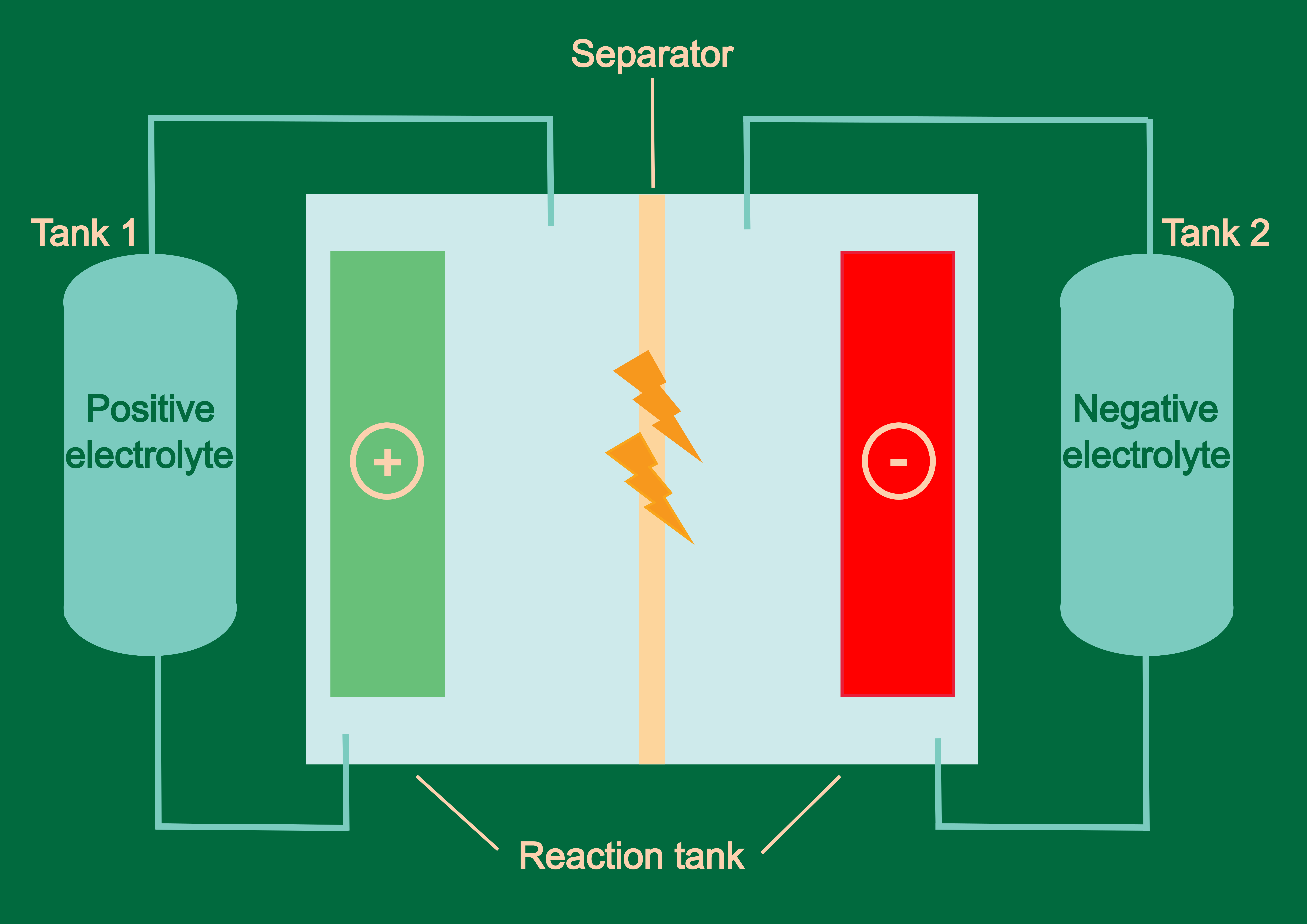
Conventional redox flow batteries contain an ion-selective membrane known as Nafion. This very durable chemical is not only expensive, but it also has serious negative environmental impacts. The new batteries that the researchers in Klosterneuburg are working on in the EU-funded “MeBattery” project do not require this separating membrane at all. To this end, the group is experimenting with various chemical substances and their ability to mix and form electro-active materials.
“In redox flow batteries, chemical species susceptible to gain or lose electrons are dissolved in electrolytes and stored in two different tanks,” Corella continues. When energy is needed, the electrolytes are pumped into a reaction chamber, where both electrolytes are separated by a membrane that allows the flow of ions but not of the dissolved chemical species. The spontaneous electrochemical reaction taking place at both sides of the membrane gives rise to the flow of electrons, therefore generating current. “By taking advantage of the physical properties of the electrolytes and playing around with their miscibility, we can get rid of the membrane. This is a breakthrough,” the young chemist adds.

From abundant elements as substitutes for rare metals to entirely new materials—with their innovative approaches, the scientists at ISTA are working on the foundations of more environmentally friendly and sustainable energy systems.